Abstract
Human adenoviruses (HAdVs) are known to cause respiratory illness outbreaks at basic military training (BMT) sites. HAdV type-4 and -7 vaccines are routinely administered at enlisted BMT sites, but not at military academies. During Aug.–Sept. 2016, U.S. Naval Academy clinical staff noted an increase in students presenting with acute respiratory illness (ARI). An investigation was conducted to determine the extent and cause of the outbreak. During 22 Aug. –11 September 2016, 652 clinic visits for ARI were identified using electronic health records. HAdV-4 was confirmed by real-time polymerase chain reaction assay in 18 out of 33 patient specimens collected and 1 additional HAdV case was detected from hospital records. Two HAdV-4 positive patients were treated for pneumonia including 1 hospitalized patient. Molecular analysis of 4 HAdV-4 isolates identified genome type 4a1, which is considered vaccine-preventable. Understanding the impact of HAdV in congregate settings other than enlisted BMT sites is necessary to inform discussions regarding future HAdV vaccine strategy.
What Are the New Findings?
In late summer 2016, the U.S. Naval Academy noted an increase in acute respiratory illness and conducted an investigation. Laboratory analysis confirmed adenovirus (HAdV-4) in 19 ill patients, and genetic sequencing confirmed a single strain of HAdV-4. This report provides new evidence of the impact of adenovirus at a federal military service academy.
What Is the Impact on Readiness and Force Health Protection?
Human adenoviruses (HAdVs) are known to cause respiratory illness outbreaks at basic military training (BMT) sites. HAdV type-4 and -7 vaccines are administered at enlisted BMT sites. Understanding the impact of HAdV in officer accession/training settings is necessary to inform discussions regarding HAdV vaccine strategy, reduce morbidity of respiratory illness in this population, and positively impact training throughout.
Background
Human adenoviruses (HAdVs) are non-enveloped double-stranded DNA viruses and are common causes of upper and lower respiratory tract illness.1 The multiple types of adenoviruses are associated with a spectrum of clinical presentations ranging from mild illness to severe disease and pneumonia.1,2 HAdV types 4 (HAdV-4) and 7 (HAdV-7) have been associated with outbreaks of acute respiratory illness (ARI) among new recruits at U.S. enlisted basic military training (BMT) sites since the 1950s.2-6 Because of evidence of multiple HAdV-4 and HAdV-7 outbreaks in enlisted BMT populations, routine vaccination against HAdV-4 and HAdV-7 at enlisted BMT sites was implemented in 1971 and resulted in a 50%–60% reduction in ARI.7
During a period of vaccine non-availability between 1999 and late 2011, rates of HAdV-related illness and hospitalizations increased dramatically at enlisted BMT sites, but rates rapidly declined following reintroduction of the vaccine in late 2011.5,8 The cessation of the HAdV-4 and HAdV-7 vaccination resulted in outbreaks of HAdV associated ARI with attack rates as high as 5%–10% per week.9-15 The HAdV-4 and HAdV-7 vaccine is a live oral vaccine approved for use in military populations 17–50 years of age.16 This vaccine is recommended by the Department of Defense (DOD) for enlisted BMT recruits and is not currently recommended for routine administration in other populations, including federal service academies, such as the U.S. Naval Academy (USNA).17 Occasionally, HAdV outbreaks have been reported in settings other than enlisted BMT sites, including colleges, inpatient and long-term care settings, and communities,18–23 but the extent and frequency of such outbreaks in the U.S. are not well described. Additionally, ARIs have previously been described as a frequent cause of morbidity at the USNA,24-25 and 1 study has implicated HAdV.25
Beginning in late Aug. 2016, staff at the USNA Brigade Medical Clinic (BMC) noted an increase in the number of students presenting with ARI including fever above 100.5°F with pharyngitis. An investigation was performed to further characterize the etiology and magnitude of this ARI outbreak.
Methods
Population and setting
The USNA is a 4-year coeducational federal service academy in Annapolis, MD. The student body is composed of approximately 4,400 students, with 1,100 students per class year; 25% of students are female. The USNA is both a military and academic training environment with living conditions comparable to civilian undergraduate institutions; all students reside in dormitory-style housing (maximum 4 persons per room), dine and socialize predominantly with members of their company (groups of approximately 150 students comprising students from all 4 class levels), and attend undergraduate courses with classmates of their year group. Students obtain non-emergency medical care at the BMC located within the dormitory facility. Dormitory-style housing at USNA with 2–4 persons per room differs substantially from Navy/Marine Corps BMT sites, where recruits are housed in open bay barracks housing 20 or more persons per room.
First-year students at USNA typically arrive on campus in early July to begin daily military training and strenuous physical conditioning prior to the start of the academic year in late Aug. Second-to-fourth-year USNA students return to campus in mid-Aug. and host joint activities for all 4 year groups immediately prior to the resumption of the academic year. In 2016, first-year students reported on 1 July, and second-to-fourth-year students returned to campus on 18 Aug. Academic courses began on 22 August 2016.
ARI surveillance
Retrospective BMC medical visit data were obtained from the Military Health Data Repository System (MDR). The MDR is a centralized data repository that captures, archives, validates, integrates, and distributes health care data from the DOD worldwide network of over 260 facilities.26 Records of clinical encounters encompassing the final 2 weeks of first-year student military training through the first 5 weeks of the academic term (8 Aug.–25 Sept. 2016) and the equivalent 7-week periods for the 2012–2015 academic years were reviewed to ascertain BMC visit numbers and rates. The combined mean of the 2012–2015 ARI visits and rates were used as the baseline. ARI visits and rates from 2016 were compared to the baseline for the equivalent 7-week period. Both initial visits and follow-up visits were included in the calculation of baseline rates. For the purposes of this investigation, an outbreak was defined as rates or visits for ARI that were more than 1.96 standard deviations above the baseline.
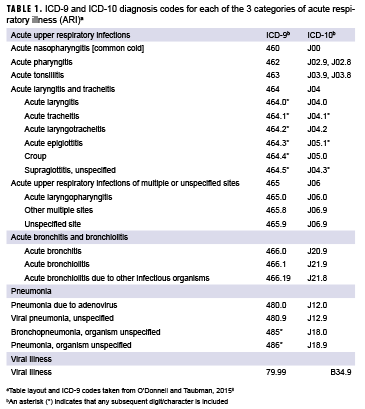
ARI was defined using a previously published standardized case definition that employed ICD-9 and ICD-10 diagnosis codes (Table 1).8 To better capture the impact of illness on clinical resources, patient visits (rather than affected students) were examined; students could be diagnosed with ARI more than once during the study timeframe. Demographic variables obtained for the 2016 patients included age, sex, company, and class year. For the period from 2012–2015, only data on ARI were accessed.
Laboratory investigations
All laboratory testing obtained was ordered by treating providers based upon clinical suspicion, recommended diagnostic protocols, and BMC clinic standard operating procedures.23 When performed, nasopharyngeal (NP) swabs were tested for influenza by Sofia Influenza A+B Fluorescent Immunoassay (FIA) (Quidel Corp., San Diego, CA). Oropharyngeal swabs (OP) were tested for Group A streptococci (GAS) by Sofia Strep A FIA (Quidel Corp).
Submission of clinical specimens for viral culture laboratory testing was based on the clinical suspicion of individual providers because early testing did not indicate that influenza or GAS were likely causative agents. Viral culture laboratory testing was initially performed at Walter Reed National Military Medical Center (WRNMMC) and then HAdV-4 positive specimens were transferred to the U.S. Centers for Disease Control and Prevention (CDC) for serotyping. Providers obtained increased numbers of samples commensurate with the initial increase in ARI cases and then later reduced the frequency of testing of patients after HAdV was identified on 9 September 2016 and was determined to be a likely cause of the increased ARI visits.
WRNMMC utilized R-Mix shell vial and D3 Ultra DFA Respiratory Virus Screening and ID Kit (Diagnostic HYBRIDS, Inc., Athens, OH) following manufacturer's recommendation with slight modifications. Briefly, specimens were inoculated onto cell monolayer in the R-Mix shell vials and stained for the presence of viral antigens using a pool of monoclonal antibodies directed against influenza A, influenza B, parainfluenza type 1, parainfluenza type 2, parainfluenza type 3, adenovirus, and respiratory syncytial virus. If virus-specific fluorescence was noted by screening, virus identification was performed using individual monoclonal antibodies staining.
At the CDC, HAdV-4 positive specimens were tested by a generic pan-HAdV real-time polymerase chain reaction (pan-rPCR) assay to confirm HAdV detection and typed by PCR and sequencing of hexon gene hypervariable regions 1-6 (HVR1-6)28 and HAdV type-specific rPCR assays.29 Specimens were also tested at the CDC for other respiratory pathogens by FTD Respiratory Pathogens 21 (FTD-21) real-time reverse transcription PCR (RT-PCR) (Fast-track diagnostics Ltd., Sliema, Malta), which tests for influenza A; influenza A (H1N1) swl; influenza B; rhinovirus; coronavirus NL63, 229E, OC43, HKU1; parainfluenza 1, 2, 3, 4; human metapneumovirus A/B; bocavirus; respiratory syncytial virus A/B; adenovirus; enterovirus; parechovirus; and Mycoplasma pneumoniae. For specimens that tested positive for both rhinovirus and enterovirus by FTD-21 rRT-PCR, RT-PCR and sequencing of partial VP4/VP2 region were performed to distinguish rhinovirus and enterovirus detection.30
CDC selected HAdV-positive specimens with sufficient volume and relatively low cycle threshold values representing patients infected at the beginning, middle, and end of the outbreak for genomic sequencing to identify similarities, and specimens were inoculated into A549 cells. For genomic sequencing, DNA libraries of the isolates were constructed using Nextera XT DNA Library Prep Kit (Illumina Inc., San Diego, CA) and paired-end sequencing was performed on the MiSeq using 500-cycle Miseq Reagent Kit V2 (Illumina Inc.). De novo assemblies were achieved using CLC Genomics Workbench v8.5.1 (CLCbio, Seoul, South Korea). Phylogenetic trees of nearly full genome sequences obtained in this study and selected from GenBank® were constructed using the neighbor-joining method implemented in MEGA7.31,32 In silico restriction enzyme analysis of the HAdV-4 genomes was performed using Geneious v8.1.6 (Biomatters Ltd. Auckland, New Zealand), and genome types were determined using established guidelines and reference fragment patterns.33,34
Case identification and clinical presentation
A confirmed HAdV case was any USNA student evaluated and diagnosed with ARI by clinical staff with symptom onset occurring during 22 Aug.–23 Sept. 2016 and a positive result for HAdV by culture/immunofluorescence assay (IFA) or rPCR of a NP or OP swab. Individual BMC electronic medical records were reviewed for all students who had a NP or OP swab submitted for HAdV testing during 22 Aug.–23 September 2016. In addition, hospital admissions records for all students admitted during 1 Aug.–30 Sept. 2016 were reviewed to account for admissions to medical facilities outside of the BMC. Navy Environmental Preventive Medicine Unit 2 clinical staff reviewed electronic health record data using a standardized questionnaire to obtain patient demographics, date of symptom onset, recent illnesses, symptom presentation, medical history, ill contacts, recent hospitalization, treatment, and radiologic findings. Medical record review and surveillance data were analyzed using Stata software, version 13.0 (StataCorp, College Station, TX) and SAS/STAT software, version 9.4 (2014, SAS Institute Inc., Cary, NC). Categorical variables were reported as percentages in each category.
Results
ARI surveillance
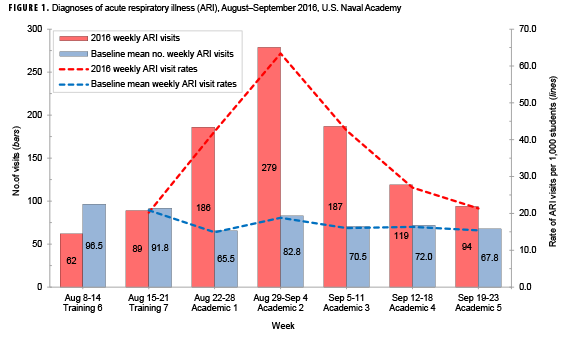
During 8 Aug.–23 Sept. 2016, 1,016 outpatient medical visits from 828 unique students (over 20% of the USNA student body) resulted in ARI diagnoses. During 22 Aug.–11 September 2016, 652 outpatient medical visits (representing 547 unique students) resulted in ARI diagnoses, a value more than 3 standard deviations greater than the baseline value. Compared to baseline visits, an excess of 433 student ARI visits occurred during this 3 week period (data not shown). Additionally, the 2016 ARI rate during the same 3-week period was 49 ARI cases per 1,000 students per week, a nearly 3-fold increase compared to the baseline rate (data not shown).
During the first academic week (22 Aug.–28 Aug. 2016), 79% (131/165) of students presenting with illness were first-year students (data not shown). Overall, 61.8% of all ARI-related visits in week 1 were for students 18 years or younger. During the second academic week (29 Aug.–4 Sept. 2016), the rate of ARI among all students (first-to-fourth-year students) peaked and was more than 3 times the baseline rate (Figure 1). In the third academic week (5 September–11 September 2016), 75.4% (141/187) of all ARI-related visits were among students 19 years or older (data not shown). Rates of ARI were similar between companies. Between 30.5% and 34.4% of the ARI patients who presented to clinic per week were female (data not shown).
Laboratory investigation
During 22 Aug.–23 Sept. 2016, clinical lab samples from ARI cases included 156 rapid GAS tests (2.6% positive) and 28 rapid influenza tests (0% positive) (data not shown). Lab samples for 33 patients were sent for viral testing, including for HAdV testing.
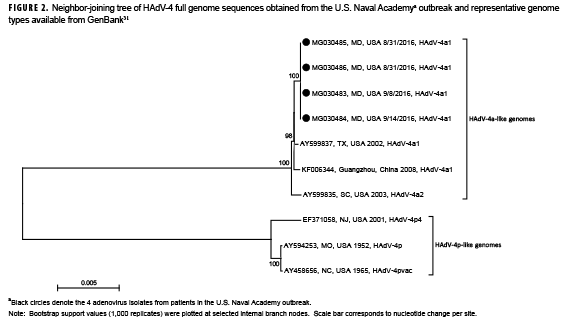
HAdV was detected by pan-rPCR in 26 of 46 (56.5%) specimens (NP: 17/33, 51.5%; OP: 9/13, 69.2%) representing 18 of the 33 patients (54.5%). Pan-rPCR detected HAdV from all culture/IFA positive samples (total: n=19 specimens, 16 patients) and from an additional 7 specimens representing 2 additional patients (total: n=26 specimens, 18 patients) who were culture/IFA negative (data not shown). Hexon HVR1–6 sequencing and HAdV-type specific rPCR identified all HAdV positive samples as HAdV-4. Genome sequences (GenBank accession number MG030483-MG030486) obtained from 4 HAdV-4 isolates were identical with each other and showed 94.3% (accession number EF3710058.2) to 99.8% (accession numbers AY599835.1, AY599837.1 and KF006344.1) nucleotide sequence similarity to other representative HAdV-4 genomes available in GenBank. Phylogenetic analysis of all sequences revealed 2 major clades, each comprising genomes exhibiting 4a-like and 4p-like restriction profiles, respectively (Figure 2). USNA sequences clustered within the 4a-like clade. Restriction profiles obtained with the USNA sequences by in silico analysis using enzymes BamHI, DraI, EcoRI, EcoRV, XhoI, and SmaI identified them as genome type 4a1.
Rhinovirus was co-detected from 1 HAdV-4 positive patient and was also detected from 4 HAdV-4 negative patients by FTD-21 rRT-PCR (data not shown). Enterovirus D68 (EV-D68) was also detected from 2 HAdV-4 negative patients by sequencing analysis. No other respiratory pathogens were identified on either rRT-PCR or culture/IFA. Nine patients were negative for all pathogens tested (data not shown).
Case identification and clinical presentation
Eighteen HAdV-4 positive cases were identified among 33 students seen at the BMC as outpatients who had specimens submitted for viral testing. Among the 33 students with specimens collected, the median number of days from symptom onset to specimen collection was 3.0 (range 1–13 days). Among 27 charts reviewed for students hospitalized from 1 Aug.–30 Sept. 2016, a single additional student was hospitalized for ARI and also tested positive for HAdV by PCR; no typing was performed on the specimen from the hospitalized student.
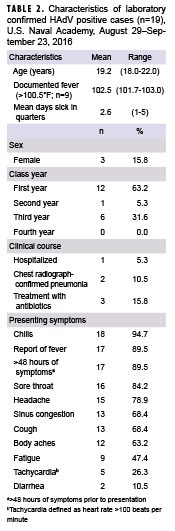
Among the 19 laboratory-confirmed cases with symptom information available, the vast majority presented clinically with 1 or more of the following: chills, reported history of fever, symptoms persisting for ≥2 days prior to presentation, sore throat, headache, sinus congestion, and cough (Table 2). Nine of the 19 HAdV positive patients had documented fever (temperature >100.5°F) determined by chart review, with a mean temperature of 102.5°F (range: 101.7–103.0°F) (Table 2). The mean number of days of sick-in-quarters (time excused from all classes and activities) was 2.6 days (range 1–5 days). There were no cases with documentation of chronic underlying illness (including asthma, heart condition, respiratory illness, diabetes, obesity, or use of a prescription medication). Two patients were diagnosed with pneumonia by chest radiograph including the single hospitalized case; chest radiographs were not obtained in 17 of the 19 cases (data not shown). No patient was placed on antivirals empirically for influenza and all patients presented with >95% oxygen saturation on room air (data not shown).
Control measures
In response to the increase in ARI cases, campus-wide control measures were implemented on 31 Aug. 2016, including 1) hand hygiene and cover-your-cough educational outreach messaging, 2) hand sanitizer station availability throughout campus, 3) strict implementation of mask use for symptomatic patients visiting the clinic, and 4) social distancing measures through sick-in-quarters restrictions for symptomatic individuals.
Editorial Comments
This investigation describes a large outbreak of respiratory illness associated with HAdV-4 that occurred among students at the USNA during Aug.–Sept. 2016. Relative to baseline, the outbreak was estimated to have contributed to over 400 excess outpatient clinic visits during this period. The clinical syndrome included high fever, chills, sore throat, headache, and cough, which frequently led to absences from scheduled activities but rarely included pneumonia or resulted in hospitalization. HAdV-4 is an important cause of ARI and conjunctivitis worldwide.35-38 Although HAdV-4 has been infrequently documented to be associated with acute respiratory illness among U.S. civilians,18,39,40 it was the predominant cause of ARI among U.S. enlisted BMT recruits before reintroduction of routine vaccination in late 2011.39,40
The 2016 outbreak occurred in a residential college-style setting among students from diverse geographic areas who reside in dormitories (2–4 persons per dorm room) and attend mass gatherings. HAdVs are noted to persist in the environment (e.g., in lockers, on bedposts, and on pillow cases) for substantial periods of time41 and to have a high degree of communicability.9 The 2016 outbreak highlights the role of HAdV as a potential cause of ARI among students residing in dormitories in college-like settings. However, it is notable that attendees of federal service academies are exposed to unique physical and military training requirements as compared with civilian undergraduate institutions.
In enlisted BMT settings, HAdV-4 infection has been linked with severe respiratory illness, with a high impact on loss of training, hospitalized days, and clinical resources.8-14 In this outbreak, cases reported a mean absence of 2.6 days following their initial visit, excluding possible follow-up visits that may have resulted in additional days of absence. Although military enlisted recruits are routinely administered the HAdV-4 and HAdV-7 vaccines, military academy students are not routinely vaccinated with the HAdV vaccine. This outbreak was associated with HAdV-4a, a type for which the existing HAdV vaccine would have potentially offered protection, as vaccination with HAdV-4p is thought to protect against symptomatic infections with HAdV-4a-like viruses.42
Li and Waddel28 first described finding 2 major genomic clusters of HAdV-4, designated here as 4p-like and 4a-like, based on restriction profile analysis, which were later confirmed and refined by whole genome sequencing.43,44 Whereas HAdV-4p-like genomes were the most prevalent in the U.S. during the late 1960s to early 1980s, HAdV-4a-like genomes came to dominate detections among military populations in later years.29 HAdV-4 strains associated with the 2016 outbreak were identified as HAdV-4a1, with sequences similar to strains circulating among U.S. military enlisted recruits in 2002 (GenBank accession number AY599837.1) and 2003 (GenBank accession number AY599835.1) and in China (GenBank accession number KF006344.1).
The investigation described here is subject to several limitations. Although HAdV-4 was identified in 18 of 33 patients with available specimens, it was not possible to quantify the proportion of the more than 400 excess ARI clinical encounters potentially associated with HAdV-4, as specimens were only collected based on the clinical suspicion of individual providers. Other respiratory viruses that likely contributed to ARI cases, including rhinovirus and EV-D68, were identified. Moreover, it is not known whether individuals with available specimens differed from individuals presenting with ARI who did not have specimens collected. Finally, risk factors for illness among identified cases were not ascertained and environmental sampling was not feasible during this investigation.
The epidemiology and burden of respiratory illness due to HAdV-4 in residential college settings and federal service academies requires further characterization. These results indicate the need for additional documentation of the impact of HAdV-4 in these settings to inform discussions of parameters for extending vaccine use beyond enlisted BMT settings. Providers and public health practitioners should consider HAdV as a potential contributor to ARI outbreaks, including those in residential campus settings.
Author Affiliations: Navy Environmental and Preventive Medicine Unit TWO (LCDR Rogers, LCDR Johnson); Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention (Ms. Lu, Ms. Killerby, Dr. Erdman, Dr. Gerber, Dr. Schneider, Dr. Watson); Naval Health Clinic, Annapolis, MD (Ms. Campbell, CDR Gallus); Walter Reed National Military Medical Center (Dr. Kamau, Ms. Froh); Navy and Marine Corps Public Health Center (Ms. Nowak); Battelle contracting agency to Respiratory Viruses Branch, Centers for Disease Control and Prevention (Dr. Sakthievel)
Acknowledgments: The authors acknowledge Tammy Servies, MD, MPH (CDR, USN, MC), Christopher Viers (HM1, USN), William Sterling (LT, USN, MSC), and The USNA Brigade Medical Clinic Team led by Cynthia Bryant, MD (CDR, USN, MC) for their support during this investigation.
Disclaimer: The content of this publication is the sole responsibility of the authors and does not necessarily reflect the views or policies of the Department of Defense (DOD), or the Departments of the Army, Navy, or Air Force, or the Centers for Disease Control and Prevention. Mention of trade names, commercial products, or organizations does not imply endorsement by the U.S. Government.
Conflicts of interest: The authors of this manuscript have no conflicts of interest to disclose.
Financial support: There are no additional sources of financial support to disclose.
References
- Baum S. Adenovirus. In: Mendell G, Bennett J, Dolin R. Principles and Practices of Infectious Diseases 8th Edition. New York: Churchill Livingstone; 2014:1787–1793.
- Foy H. Adenoviruses. In: Evans A, Kaslow R. Viral Infections of Humans: Epidemiology and Control. New York: Plenum Medical Book Company; 1997:119–138.
- Dudding BA, Top FH, Jr., Winter PE, Buescher EL, Lamson TH, Leibovitz A. Acute respiratory disease in military trainees: The adenovirus surveillance program, 1966–1971. Am J Epidemiol. 1973;97(3):187–198.
- Hoke CH, Jr., Snyder CE, Jr. History of the restoration of adenovirus type 4 and type 7 vaccine, live oral (adenovirus vaccine) in the context of the Department of Defense acquisition system. Vaccine. 2013;31(12):1623–1632.
- Gray GC, Callahan JD, Hawksworth AW, Fisher CA, Gaydos JC. Respiratory diseases among U.S. military personnel: Countering emerging threats. Emerg Infect Dis. 1999;5(3):379–385.
- Top FH, Jr., Dudding BA, Russell PK, Buescher EL. Control of respiratory disease in recruits with types 4 and 7 adenovirus vaccines. Am J Epidemiol. 1971;94(2):142–146.
- Top FH. Control of adenovirus acute respiratory disease in U.S. Army trainees. Yale J Biol Med. 1975;48(3):185-195.
- O'Donnell FL, Taubman SB. Follow-up analysis of the incidence of acute respiratory infections among enlisted service members during their first year of military service before and after the 2011 resumption of adenovirus vaccination of basic trainees. MSMR. 2015;22(12):2–7.
- Kolavic-Gray SA, Binn LN, Sanchez JL, et al. Large epidemic of adenovirus type 4 infection among military trainees: Epidemiological, clinical, and laboratory studies. Clin Infect Dis. 2002;35(7):808–818.
- Hendrix RM, Lindner JL, Benton FR, et al. Large, persistent epidemic of adenovirus type 4-associated acute respiratory disease in U.S. Army trainees. Emerg Infect Dis. 1999;5(6):798–801.
- Barraza EM, Ludwig SL, Gaydos JC, Brundage JF. Reemergence of adenovirus type 4 acute respiratory disease in military trainees: Report of an outbreak during a lapse in vaccination. J Infect Dis. 1999;179(6):1531–1533.
- Gray GC, Goswami PR, Malasig MD, et al. Adult adenovirus infections: Loss of orphaned vaccines precipitates military respiratory disease epidemics. For the Adenovirus Surveillance Group. Clin Infect Dis. 2000;31(3):663–670.
- McNeill KM, Ridgely Benton F, Monteith SC, Tuchscherer MA, Gaydos JC. Epidemic spread of adenovirus type 4-associated acute respiratory disease between U.S. Army installations. Emerg Infect Dis. 2000;6(4):415–419.
- Ryan MA, Gray GC, Smith B, McKeehan JA, Hawksworth AW, Malasig MD. Large epidemic of respiratory illness due to adenovirus types 7 and 3 in healthy young adults. Clin Infect Dis. 2002;34(5):577–582.
- Coldren RL, Feighner B, DuVernoy T, Jordan N, Gonzalez R, Alsip B. Adenovirus type 4 outbreak among basic trainees, Ft. Benning, Georgia, April-May 2000. MSMR. 2000;6(6):2–7.
- Center for Biologics Evaluation and Research. Approved Products—Adenovirus Type 4 and Type 7 Vaccine, Live, Oral. U.S. Food and Drug Administration. https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM247515.pdf. Accessed on 01 April 2017.
- Immunizations and Chemoprophylaxis for the Prevention of Infectious Diseases: Army Regulation 40–562 BUMEDINST 6230.15B AFI 48–110 IP CG COMDTINST M6230.4G. In: Departments of the Army, the Navy, the Air Force, and the Coast Guard, 2013.
- Sivan AV, Lee T, Binn LN, Gaydos JC. Adenovirus-associated acute respiratory disease in healthy adolescents and adults: A literature review. Mil Med. 2007;172(11):1198–203.
- Yang X., Wang Q, Liang B, et al. An outbreak of acute respiratory disease caused by a virus associated RNA II gene mutation strain of human adenovirus 7 in China, 2015. PLoS One. 2017;12(2):e0172519
- Tsou TP et al. Community outbreak of adenovirus, Taiwan, 2011. Emerg Infect Dis. 2012;18(11):1825–1832.
- Kajon AE, Lamson DM, Blair CR, et al. Adenovirus type 4 respiratory infections among civilian adults, northeastern United States, 2011–2015. Emerg Infect Dis. 2018;24(2):201-209.
- Scott MK, Chommanard C, Lu X, et al. Human adenovirus associated with severe respiratory infection, Oregon, USA, 2013–2014. Emerg Infect Dis. 2016;22:1044.
- Kandel R, Srinivasan A, D'Agata EM, Lu X, Erdman D, Jhung M. Outbreak of adenovirus type 4 infection in a long-term care facility for the elderly. Infect Control Hosp Epidemiol. 2010;31(7):755–757.
- Blakenship, TL, Gackstetter GD, Gray, GC. History of respiratory illness at the U.S. Naval Academy. Mil Med. 2001;166(7):581–586.
- Gray GC, Schultz RG, Gackstetter GD, et al. Prospective study of respiratory infections at the U.S. Naval Academy. Mil Med. 2001;166(9):759–763.
- Defense Health Agency. Military Health System Data Repository (MDR). 2013. Available at: https://health.mil/Military-Health-Topics/Technology/Clinical-Support/Military-Health-System-Data-Repository. Accessed on 13 July, 2017.
- Pelucchi C, Grigoryan L, Galeone C, et al. Guideline for the management of acute sore throat. Clin Microbiol Infect. 2012;18 Suppl 1:1–28.
- Lu X, Erdman DD. Molecular typing of human adenoviruses by PCR and sequencing of a partial region of the hexon gene. Arch Virol. 2006;151(8):1587–1602.
- Lu X, Trujillo-Lopez E, Lott L, Erdman DD. Quantitative real-time PCR assay panel for detection and type-specific identification of epidemic respiratory human adenoviruses. J Clin Microbiol. 2013;51(4):1089–1093.
- Lu X, Schneider E, Jain S, et al. Rhinovirus viremia in patients hospitalized with community acquired pneumonia. J Infect Dis. 2017;216(9):1104–1111.
- Benson DA, Cavanaugh M, Karsch-Misrachi I, Lipman DJ, Ostell J, Sayers EW. GenBank. Nucleic Acids Res. 2017;45(D1):D37-D42.
- Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol. 2016;33(7):1870-1874.
- Li QG, Wadell G. The degree of genetic variability among adenovirus type 4 strains isolated from man and chimpanzee. Arch Virol. 1988;101(1-2):65–77.
- Kajon AE, Moseley JM, Metzgar D, et al. Molecular epidemiology of adenovirus type 4 infections in U.S. military recruits in the postvaccination era (1997–2003). J Infect Dis. 2007;196(1):67–75.
- Chen HL, Chiou SS, Hsiao HP, et al. Respiratory adenoviral infections in children: A study of hospitalized cases in Southern Taiwan in 2001–2002. J Trop Pediatr. 2004;50(5):279–284.
- Ariga T, Shimada Y, Ohgami K, et al. New genome type of adenovirus serotype 4 caused nosocomial infections associated with epidemic conjunctivitis in Japan. J Clin Microbiol. 2004;42(8):3644–3648.
- Schepetiuk SK, Norton R, Kok T, Irving LG. Outbreak of adenovirus type 4 conjunctivitis in South Australia. J Med Virol. 1993;41(4):316–318.
- Cooper RJ, Bailey AS, Killough R, Richmond SJ. Genome analysis of adenovirus 4 isolated over a six year period. J Med Virol. 1993;39(1):62–66.
- Hierholzer JC. Adenoviruses. In: Lennette EH, Lennette DA, Lennette ET. Diagnostic Procedures for Viral, Rickettsial, and Chlamydial Infections, 7th ed Washington DC: American Public Health Association, 1995:169–188.
- Radin JM, Hawksworth AW, Blair PJ, et al. Dramatic decline of respiratory illness among US military recruits after the renewed use of adenovirus vaccines. Clin Infect Dis. 2014;59(7):962–968.
- Russell KL, Broderick MP, Franklin SE, et al. Transmission dynamics and prospective environmental sampling of adenovirus in a military recruit setting. J Infect Dis. 2006;194(7):77–85.
- Kuschner RA, Russell KL, Abuja M, et al. A phase 3, randomized, double-blind, placebo-controlled study of the safety and efficacy of the live, oral adenovirus type 4 and type 7 vaccine, in U.S. military recruits. Vaccine. 2013;31(28):2963–2971.
- Hang J, Vento TJ, Norby EA, et al. Adenovirus type 4 respiratory infections with a concurrent outbreak of coxsackievirus A21 among United States Army Basic Trainees, a retrospective viral etiology study using next-generation sequencing. J Med Virol. 2017;89(8):1387–1394.
- Dehghan S, Seto J, Liu EB, et al. Computational analysis of four human adenovirus type 4 genomes reveals molecular evolution through two interspecies recombination events. Virology. 2013;443(2):197–207.