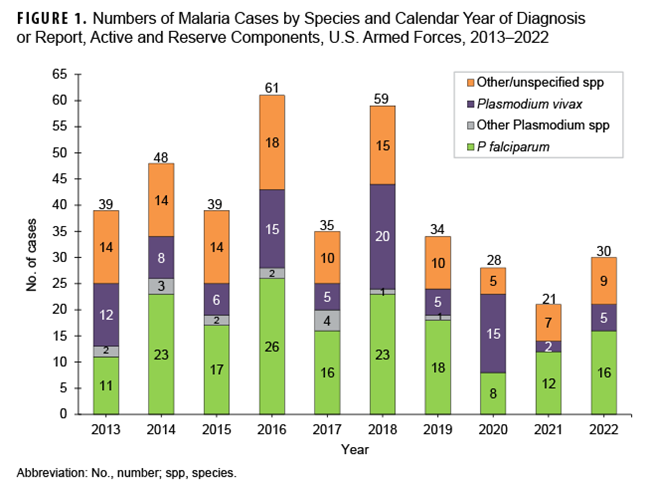
Malaria infection remains a potential health threat to U.S. service members located in or near endemic areas due to duty assignment, participation in contingency operations, or personal travel. In 2022, a total of 30 active and reserve component service members were diagnosed with or reported to have malaria, a 42.9% increase from the 21 cases identified in 2021. Over half of the malaria cases in 2022 were caused by Plasmodium falciparum (53.3%; n=16) and one-sixth (16.7%; n=5) were attributed to P vivax. The remaining 9 cases were associated with other or unspecified types of malaria. Malaria cases were diagnosed or reported from 19 different medical facilities—15 in the U.S. and 1 each from Germany, Africa, South Korea, and Japan. Of the 28 cases with a known location of diagnosis, 9 (32.1%) were reported from or diagnosed outside the U.S.
What are the new findings?
Total malaria cases have decreased since 2016, likely due to the reduction in forces deployed to Afghanistan, a known malaria risk area. The reduction in case counts over the last three years of the period may be due in part, to COVID-19-related travel restrictions, curtailing travel to malaria risk areas.
What is the impact on readiness and force health protection?
Malaria infection causes acute incapacitation. P falciparum malaria poses a high risk of serious sequelae including death. Unprotected forces can experience extraordinarily high attack rates in highly endemic areas, potentially causing mission failure. Malaria poses a risk for service members deployed to endemic regions as well as those traveling to such areas for personal reasons. The finding that P falciparum malaria was diagnosed in more than half of cases in 2022 underscores the need for continued emphasis on effective preventive measures against this most dangerous malaria strain.
Background
Since 1999, the MSMR has published regular updates on malaria incidence among U.S. service members.1-3 This year's update employs methods similar to previous analyses describing the epidemiologic patterns of malaria incidence among service members in the active and reserve components of the U.S. Armed Forces. The MSMR's focus on malaria reflects both historical lessons about this mosquito-borne disease and its continuing threat to military operations and service members’ health.
Malaria is a febrile parasitic disease transmitted through the bite of an infected female Anopheles mosquito. The parasite P falciparum is responsible for the deadliest form of malaria and is most prevalent in Africa.4 P vivax is the most widely distributed parasite species, with relatively high infection prevalence in the Western Pacific, Southeast Asia, and Eastern Mediterranean regions and less densely populated areas of the Americas.4
P vivax and P falciparum have distinct epidemiologies. A major difference is P vivax's ability to cause relapses weeks or months following primary infection due to its activation of hypnozoites, dormant liver-stage parasites.5 This infection reservoir allows P vivax's survival during mosquito-free cold seasons, expanding its geographic range far into temperate zones such as the Korean peninsula.6 Relapse can be prevented with presumptive anti-relapse therapy (PART), but the risk must be recognized, with appropriate therapy (primaquine or tafenoquine) prescribed and regimen successfully completed after departing the malaria-endemic area.7,8
In 2021, approximately 95% of global malaria cases and related deaths were in sub-Saharan Africa, where four countries accounted for more than half (52%) of this mortality: Nigeria (31%), Democratic Republic of the Congo (13%), the Niger (4%), and the United Republic of Tanzania (4%).4 Most of these cases and deaths were due to mosquito-transmitted P falciparum among children under age 5.4 In 2021, 2% of global estimated malaria cases were caused by P vivax.4 While heightened malaria-control efforts have reduced the incidence of P falciparum malaria in many areas, the proportion of P vivax malaria cases has increased in some regions where both parasites are present.9,10
Methods
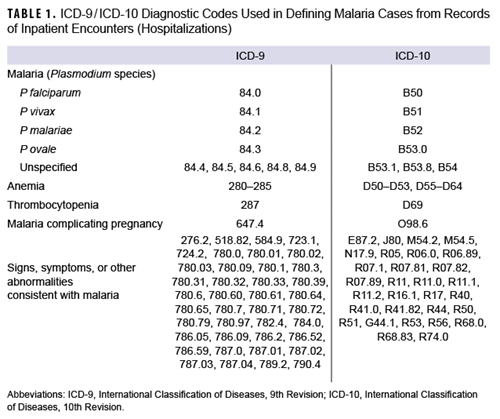
The surveillance period for this report was January 1, 2013 through December 31, 2022. The surveillance population included Army, Navy, Air Force, and Marine Corps active and reserve component members of the U.S. Armed Forces. The records of the Defense Medical Surveillance System were searched for qualifying evidence of malaria diagnoses from reportable medical events, hospitalizations or outpatient encounters (in military and non-military facilities), and laboratory results generated at military facilities. The case definition criteria included 1) an RME record of confirmed malaria, 2) a hospitalization record with a primary diagnosis of malaria, 3) a hospitalization record with a nonprimary diagnosis of malaria due to a specific Plasmodium species, 4) a hospitalization record with a nonprimary diagnosis of malaria and diagnosis of anemia, thrombocytopenia and related conditions, or malaria complicating pregnancy in any diagnostic position, 5) a hospitalization record with a nonprimary diagnosis of malaria plus diagnoses of signs or symptoms consistent with malaria in each diagnostic position antecedent to malaria,11 or 6) a positive malaria antigen test plus an outpatient record with a diagnosis of malaria in any diagnostic position within 30 days of the specimen collection date. The relevant International Classification of Diseases, 9th and 10th Revision (ICD-9 and ICD-10, respectively) codes used to identify cases are shown in Table 1.
The analysis restricted each service member to one episode of malaria per 365-day period. When multiple records documented a single episode, the date of the earliest record was considered the date of clinical onset. Records within 30 days of the clinical onset date were reviewed for evidence of a Plasmodium species.
Presumed location of malaria acquisition was estimated using a hierarchical algorithm: 1) cases diagnosed in a malaria-endemic country were considered acquired there, 2) RMEs listing exposures to malaria-endemic locations were considered acquired in those locations, 3) RMEs not listing exposures to malaria-endemic locations but reported from installations in such locations were considered acquired there, 4) cases diagnosed among service members during or within 30 days of deployment or assignment to a malaria-endemic country were considered acquired in that country, and 5) cases diagnosed among service members deployed or assigned to a malaria-endemic country within two years before diagnosis were considered acquired in those respective countries. All remaining cases were considered acquired in unknown locations.
Results
In 2022, a total of 30 U.S. service members were diagnosed with or reported to have malaria (Table 2), resulting in a rate of 1.4 per 100,000 persons (data not shown).
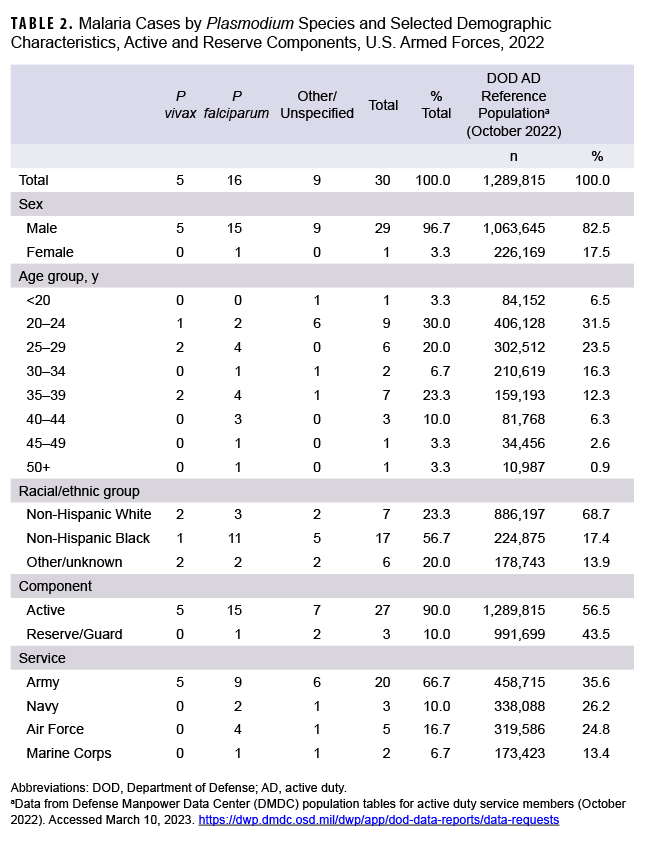
The 2022 total is a 42.9% increase from the 21 cases ascertained in 2021 (Figure 1). Twenty-two of the 30 cases (73.3%) in 2022 were identified from inpatient data and reported as RMEs; the remaining eight cases were identified from inpatient data and did not have associated RMEs. In 2022, no cases were identified from laboratory data in combination with an outpatient record of malaria.
As in previous years, in 2022 the majority of U.S. military members diagnosed with malaria were men (96.7%), members of the active component (90.0%), and in the Army (66.7%). In 2022, non-Hispanic Black service members and those under age 30 accounted for the most cases of malaria (56.7% and 53.3%, respectively) (Table 2).
Over half of the malaria cases in 2022 were caused by P falciparum (53.3%; n=16). Of the 14 cases not attributed to P falciparum, 5 (16.7%) were identified as due to P vivax and 9 were labeled as associated with other/unspecified types of malaria (30.0%) (Figure 1). This result reflects historical data over a 10-year surveillance period, where malaria cases caused by P falciparum have accounted for the largest number of cases (n=170; 43.1%), followed by other/unspecified species (n=116; 29.4%), P vivax (n=93; 23.6%), and other Plasmodium species (n=15; 3.8%). The annual percentages of cases attributed to P vivax showed the greatest variability during the period, ranging from 9.5% in 2021 to 53.6% in 2020.
During the 2013 to 2022 surveillance period, malaria cases attributed to Africa accounted for the greatest number of cases (n=149; 37.8%), followed by other/unspecified locations (n=96; 24.4%), Korea (n=67; 17.0%), Afghanistan (n=80; 9.6%), and South/Central America (n=2; 0.5%) (Figure 2).
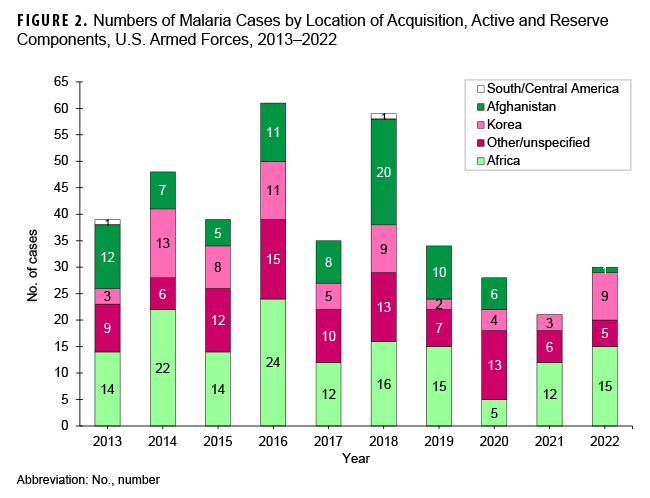
The annual percentages of cases associated with Africa had the greatest variability during the 10-year surveillance period, ranging from 17.9% in 2020 to 57.1% in 2021. From 2021 to 2022, the number of cases associated with Korea demonstrated the greatest increase, from 3 to 9; these 9 cases are the most from Korea since 2018. Since 2013, only 2 years (2014, with 13 cases; and 2016, with 11 cases) recorded higher totals (Figure 2). Malaria cases were diagnosed or reported in 2022 at 19 different medical facilities in the U.S., Germany, Africa, South Korea, and Japan (Table 3).
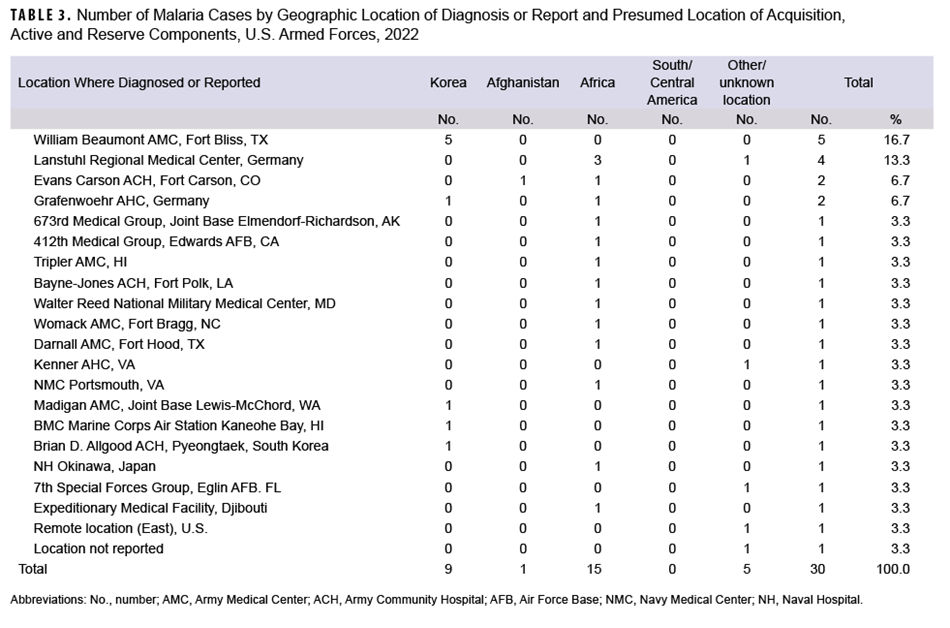
Most cases acquired in Africa were caused by P falciparum (80.0%;12/15), and more than two-fifths (44.4%; 4/9) of the cases considered as acquired in Korea were caused by P vivax (Figure 3). Of the 15 malaria infections in 2022 considered to be acquired in Africa, 4 were linked to Nigeria; 4 were associated with unknown African locations; 3 were linked to Niger; and 1 each were linked to Chad, Cameroon, Djibuti, and Uganda (data not shown). Examination of 22 malaria case records reported as RMEs revealed that 12 of the exposures were classified as deployment-related while 10 were categorized as nonduty-related, of which nine were considered acquired in Africa. Non-Hispanic Black service members accounted for eight of those nonduty cases; leisure travel to African countries was documented in the RME records of four of these service members.
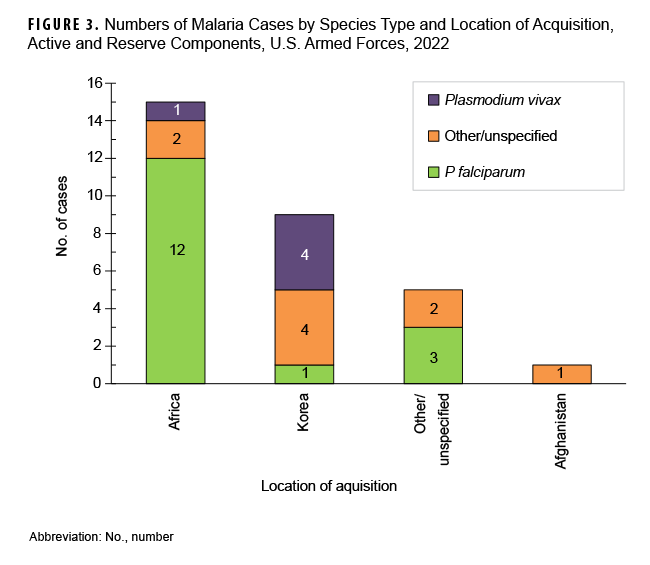
From 2013 to 2022, most non-P vivax malaria cases (70.2%) were diagnosed or reported during the six months from the middle of spring through the middle of autumn (May-October) in the Northern Hemisphere (Figure 4). This result is comparable to 2022, when 72.0% (n=23) of non-P vivax malaria cases among U.S. service members were diagnosed from May through October (data not shown). During the 10-year surveillance period, the proportions of non-P vivax malaria cases diagnosed or reported from May to October varied by region of acquisition: Korea (87.1%; 27/31); Afghanistan (86.8%; 33/38); Africa (68.3%; 97/142); and South/Central America (100.0%; 1/1) (data not shown).
Discussion
In 2022 P falciparum was responsible for more than half of U.S. service member malaria cases, underscoring the need for continued emphasis on prevention of this disease, given its potential severity and risk of death. Adherence to malaria prevention protocols is critical for military personnel when entering malaria-endemic areas. Personal protective measures against malaria include bed nets, topical insect repellent, permethrin-treated uniforms, along with chemoprophylaxis regimen compliance. Current Department of Defense guidance on medications for malaria prophylaxis summarizes the roles of chloroquine, atovaquone-proguanil, doxycycline, mefloquine, primaquine, and tafenoquine.12
Malaria remains a potential health threat to U.S. military personnel in endemic areas as a consequence of long-term duty assignments, shorter-term contingency operations, as well as personal travel. The low case counts and rate of malaria reported during the surveillance period are primarily due to the limited number of service members at risk, as most duty locations in malaria-endemic countries are far removed from areas of malaria transmission. Complacency and inattention to the serious threat of malaria, however, can have disastrous consequences during deployment to malaria-endemic regions. Just 2 decades ago, in 2003, 225 U.S. Marines were sent to augment U.S. embassy security in Liberia,13 and that mission’s failure of appropriate countermeasures led to 80 suspected cases of P falciparum malaria, with an estimated attack rate of 36%. Those malaria cases necessitated 44 medical evacuations and resulted in 5 complicated cases requiring ICU admission, ventilator, and vasopressor support.13
In addition to deployment exposures, malaria poses a significant medical concern for service members on leave traveling to malaria-endemic regions14,15; of particular concern is foreign-born personnel who travel on personal leave to their countries of origin. A prior study demonstrated that malaria rates among military members born in 7 western Africa countries were 44 times greater than those born in the U.S.14 Leisure travel to specific African countries as reported on RME records may account, at least in part, for the disproportionately high malaria rates observed among non-Hispanic Black service members in this report. Among those service members visiting their birth countries in malaria-endemic regions, susceptibility due to loss of partial immunity from previous continuous exposure poses a substantial risk for infection and morbidity.16
The observations about diagnosis seasonality for non-P vivax malaria are compatible with the presumption that risk of acquiring malaria in a temperate climatic zone of the Northern Hemisphere is greatest from May through October. Given the typical incubation periods of malaria infection (approximately 9-14 days for P falciparum, 12-18 days for P vivax and P ovale, and 18-40 days for P malariae)17 and the seasonal disappearance of biting mosquitoes during the winter, most malaria acquired in Korea and Afghanistan would be expected to cause initial symptoms during the warmer months. If primary prophylaxis is taken during the exposure period, however, initial symptoms may be suppressed. Studies of P vivax malaria in Korea have found that duration between primary infection and relapse among different P vivax strains ranges between 8 days and 8-13 months.18 Up to 40–50% of infected individuals do not manifest symptoms until 6-11 months after infection.19 Transmission of malaria in tropical regions such as sub-Saharan Africa occurs year-round but is location-specific.20
Much of the decline in cases early in the surveillance period is attributable to the decreases in military personnel in Afghanistan. The lower case counts in the last 3 years of the period may be due, at least in part, to COVID-19-related travel restrictions to, from, or through many malaria-endemic areas.
There are limitations to this report that should be considered when interpreting its findings. Ascertainment of malaria cases, especially among reserve components as well as non-deployment-related exposures, is likely incomplete, leading to rate underestimation. Some cases treated at deployed or non-U.S. military medical facilities may not have been reported or otherwise ascertained at the time of analysis. It should be noted that medical data from July 2017 through October 2019 at sites using the Military Health System's new electronic health record, MHS GENESIS, are not available in the DMSS—these sites include Naval Hospital Oak Harbor, Naval Hospital Bremerton, Air Force Medical Services Fairchild, and Madigan Army Medical Center. Medical encounter data for individuals seeking care at any of those facilities from July 2017 through 2019 were not included in the current analysis.
Diagnoses of malaria documented only in outpatient settings without confirmatory testing and not reported as notifiable events were not included in this report. The geographic location where malaria was acquired was estimated from reported information. Some cases had reported exposures in multiple malaria-endemic regions or areas, and others had no relevant exposure or information. Personal travel to or deployment in malaria-endemic countries was not accounted for unless specified in notifiable event reports. Limited information on species type in RME records highlights the need for more attention to complete documentation of reportable conditions.
Acknowledgements
The authors thank the Navy Marine Corps Public Health Center, Portsmouth, VA, for providing laboratory data for this analysis.
References
- U.S. Army Center for Health Promotion and Preventive Medicine. Malaria, U.S. Army, 1998. MSMR. 1999;5(1):2-3.
- U.S. Army Center for Health Promotion and Preventive Medicine. Malaria experience among U.S. active duty soldiers, 1997-1999. MSMR. 1999;5(8)2-3.
- Armed Forces Health Surveillance Branch. Update: malaria, U.S. Armed Forces, 2021. MSMR. 2022;27(2):2-7.
- World Health Organization. World Malaria Report 2022. Geneva, Switzerland: World Health Organization; 2022. Accessed January 23, 2023. https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2022
- Howes RE, Battle KE, Mendis KN, Smith DL, Cibulskis RE, Baird JK, Hay SI. Global epidemiology of Plasmodium vivax. Am J Trop Med Hyg. 2016;95(suppl 6):15-34.
- Gething PW, Elyazar IR, Moyes, et al. A long neglected world malaria map: Plasmodium vivax endemicity in 2010. PLoS Negl Trop Dis. 2012;6(9):e1814.
- Nascimento TL, Vasconcelos SP, Peres Y, Oliveira MJS, Taminato M, Souza KMJ. Prevalence of malaria relapse: systematic review with meta-analysis. Rev Latino-Am Enfermagem. 2019;27:e3111.
- Haston JC, Hwang J, Tan KR. Guidance for using tafenoquine for prevention and antirelapse therapy for malaria–United States, 2019. MMWR Morb Mortal Wkly Rep. 2019;68(46):1062-1068.
- Price RN, Commons RJ, Battle KE, Thriemer K, Mendis K. Plasmodium vivax in the era of the shrinking P. falciparum map. Trends Parasitol. 2020;36(6):560-570.
- Battle KE, Lucas TC, Nguyen M, et al. Mapping the global endemicity and clinical burden of Plasmodium vivax, 2000–17: a spatial and temporal modelling study. Lancet. 2019;394(10195):332-343. Armed Forces Health Surveillance Branch. Surveillance Case Definition: Malaria. February 2019. Accessed February 24, 2023. //Reference-Center/Publications/2014/12/01/Malaria.
- Defense Health Agency. Procedural Instruction 6490.03. Deployment Health Procedures. December 17, 2019.
- Whitman TJ, Coyne PE, Magill AJ, et al. An outbreak of Plasmodium falciparum malaria in U.S. Marines deployed to Liberia. Am J Trop Med Hyg. 2010;83(2):258-265.
- Wertheimer ER, Brundage JF, Fukuda MM. High rates of malaria among US military members born in malaria-endemic countries, 2002–2010. Emerg Infect Dis. 2011;17(9):1701-1703.
- Ashley DP, et al. for the IDCRP TravMil study group. A comparison of pretravel health care, travel-related exposures, and illnesses among pediatric and adult U.S. Military beneficiaries. Am J Trop Med Hyg. 2019;100(5):1285-1289.
- Pousibert-Puerto J, Lozano-Serrano AB, Soriano-Perez MJ, et al. Migration-associated malaria from Africa in southern Spain. Parasites Vectors. 2021;14(240).
- White NJ. Malaria. In: Cook GC, Zumla AI, eds. Manson’s Tropical Diseases. 22nd ed. London: Saunders Elsevier; 2009:1201-1300.
- White, NJ. Determinants of relapse periodicity in Plasmodium vivax malaria. Malar J. 2011;(10):297.
- Distelhorst JT, Marcum RE, Klein TA, Kim HC, Lee WJ. Report of two cases of vivax malaria in U.S. soldiers and a review of malaria in the Republic of Korea. MSMR. 2014;21(1):8-14.
- Fairhurst RM, Wellems TE. Plasmodium species (malaria). In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010.